Cell & Gene Therapy (CGT) CDMO Market is anticipated to grow at a CAGR of ~25% to reach ~$5 billion by 2025
Robust CGT pipeline with rising number of product approvals, strong support from large pharma and biotech, and consistent investor enthusiasm (~$20 bn raised in 2020) is set to increase the demand for CGT services. An increasing pipeline of therapies are nearing regulatory decisions. Out of 1,220 ongoing clinical trials in 2020, 152 were in Phase 3 — FDA and EMA expect approval of 10-20 cell and gene therapies each year by 2025.
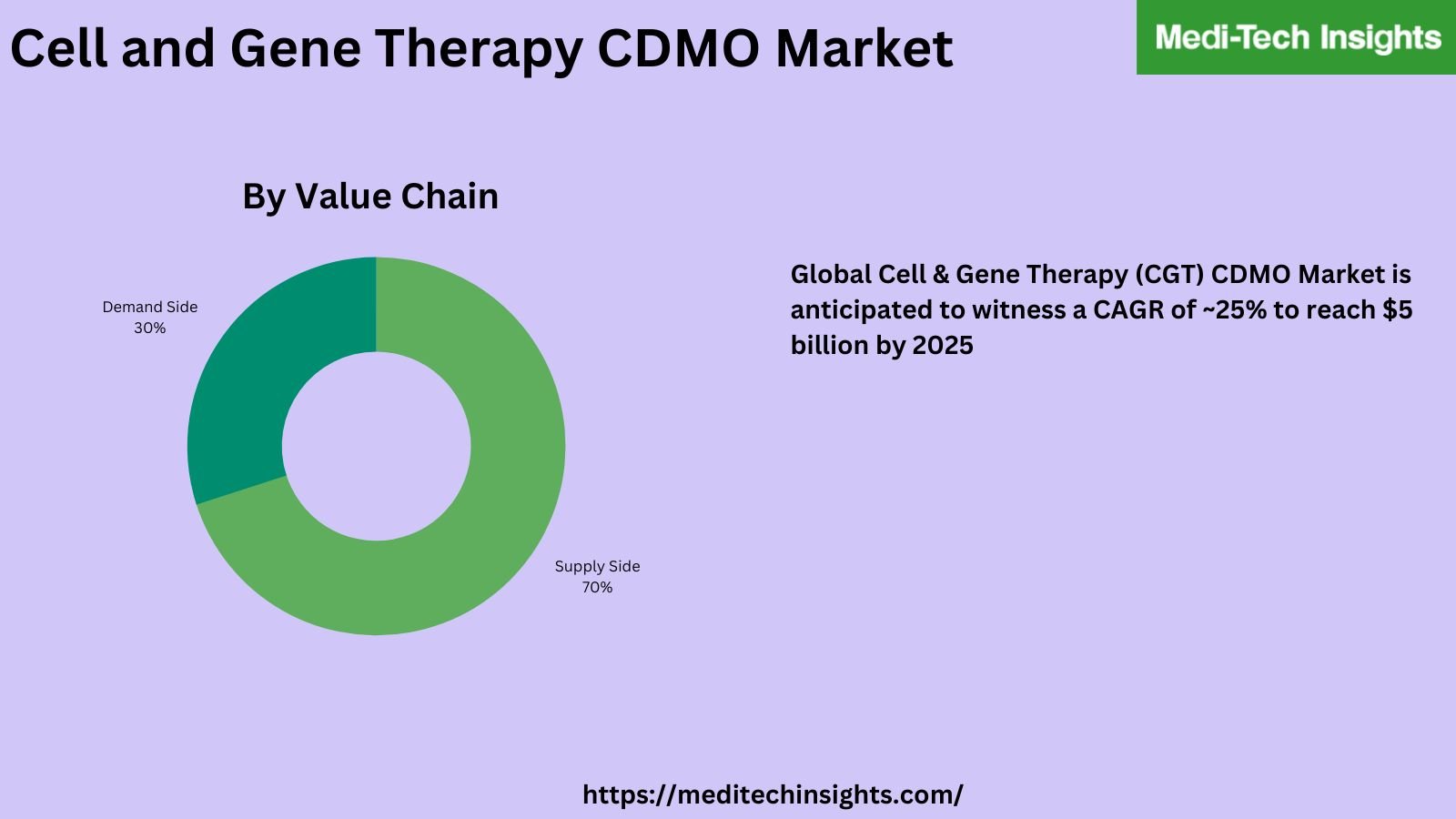
The global cell & gene therapy (CGT) CDMO market is anticipated to witness a CAGR of ~25% to reach ~$5 billion by 2025. This growth will be fueled by the rising incidence of cancer and other targeted diseases, rapidly expanding research on CGTs, substantial funding by VCs and technological innovations.
Growing Demand for Full-service/One-stop-shop CDMOs
Full-service CDMOs that can support with both development and manufacturing are in utmost demand. Recently, smaller biotech companies or research universities that rely on CDMOs for everything preclinical development to packaging have accounted for a large share of CGT development.
“There is increasing demand for Cell and Gene Therapy CTDMOs who can offer integrated development, manufacturing, and testing services. For CGT companies, outsourcing manufacturing and testing operations to a capable CTDMO can reduce development timelines, provide supplementary capacity, and ultimately control costs.” - Senior Director, Tier 1 CGT CDMO, US
Explore Premium Report on Cell & Gene Therapy (CGT) CDMO Market @ https://meditechinsights.com/cell-and-gene-therapy-cdmo-market/
Significant Outsourcing in CGT Industry due to Lack of Internal Capabilities
The level of outsourcing in CGT now approaches 60%, which is far higher than the level observed in the entire pharma/biologics industry. A high level of expertise is necessary in the complex field of cell & gene therapy. Also, it has costly production and different manufacturing process/infrastructure requirements as compared to traditional small molecules.
Big pharma currently lacks production-ready infrastructure and are increasingly looking for CGT CDMOs to enter/expand their presence in fast-growing CGT market. Additionally, the increasing number of small biotech’s entering the market with limited manufacturing capabilities and/or lack of expertise presents CGT CDMOs with additional growth opportunities.
North America is the Largest and Fastest Growing Region for Cell & Gene Therapy (CGT) CDMO Market
North America is projected to witness fastest growth and continue its dominance in the coming 5 years owing to growing manufacturing capacity for CGTs, favorable regulatory approval process, growing CGT approvals per year, and strong product pipeline of CGTs in the U.S.
Competitive Landscape Analysis of Cell & Gene Therapy (CGT) CDMO Market
The cell & gene therapy (CGT) CDMO market is marked by presence of both established players and several small and mid-sized players. Some of the leading players include Lonza, Catalent, Patheon, Wuxi Advanced Therapies and Charles River Laboratories. Other promising players include Vive Biotech, SK Bioscience, Fujifilm, Vibalogics, Exothera, Takara, Oxford Biomedica, Viralgen and AGC Biologics, among several others.
For More Detailed Insights, Contact Us @ https://meditechinsights.com/contact-us/
About Medi-Tech Insights
Medi-Tech Insights is a healthcare-focused business research & insights firm. Our clients include Fortune 500 companies, blue-chip investors & hyper-growth start-ups. We have completed 100+ projects in Digital Health, Healthcare IT, Medical Technology, Medical Devices & Pharma Services.
Contact:
Ruta Halde
Associate, Medi-Tech Insights
+32 498 86 80 79
info@meditechinsights.com


 Tejashri
Tejashri 









